- What is Prostate Cancer (Neuroendocrine Carcinoma of the Prostate)?
- Statistics
- Risk Factors
- Progression
- Symptoms
- Clinical Examination
- How is it Diagnosed
- Prognosis
- Treatment
- References
What is Prostate Cancer (Neuroendocrine Carcinoma of the Prostate)?
The prostate is a gland found only in men. It surrounds the urethra (the tube through the penis that drains urine from the bladder), and sits at the base of the bladder. It is normally about the size of a walnut. The function of the prostate is to produce fluid that forms a component of semen. The prostate is made up of mainly glandular tissue; however, there are also some specialised epithelial (lining) cells called neuroendocrine cells. These cells secrete hormones, which act as chemical messengers in the body.
Cancer occurs when an abnormality occurs in the genes of a cell, causing it to multiply and interfere with the normal function of a tissue. Once the cancer reaches a certain size, the abnormal cells can spread to other parts of the body and causes cancerous tumours to grow there (this is known as metastasis). If a tumour is capable of spreading to other parts of the body in this way, it is called malignant.
If cancer arises in the neuroendocrine cells of the prostate, it is known as neuroendocrine carcinoma of the prostate. This is a rare form of prostate cancer.
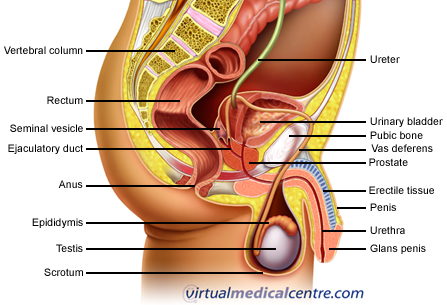
Statistics
Prostate cancer is the sixth most common cancer in the world and the most common form of cancer in Australian men (apart from melanomas). Australia also has the highest incidence of prostate cancer in the world (105 cases per 100,000 males).
The mean age at diagnosis is 68.4 years old and 85% of new cases occur in men over 60.In men of all ages, 85% will live for 5 years or longer from the time of diagnosis.To age 85, a quarter of Australian men will be diagnosed with prostate cancer and 1 in 25 will die from it, making it the second leading cause of cancer death in Australia.
Although prostate cancer is the second most common cause of cancer in Aboriginal Australian males (after lung cancer), the rates are lower in Indigenous men than in non-Indigenous men. However, it is not clear whether Indigenous Australians are truly less likely to develop prostate cancer, or whether lower rates of screening and diagnostic testing as well as a shorter life expectancy are contributing factors. There has been a significant increase in the rates of diagnosed prostate cancer since the screening of asymptomatic men became popular in the 1990s.
Primary neuroendocrine carcinoma is very rare and accounts for less than 5% of prostate cancers. More frequently, small sections of neuroendocrine carcinoma occur within the far more common adenocarcinoma form. This occurs because cancer cells are rapidly changing and dividing and tend to change their form.
Risk Factors

Age
The main risk factor for prostate cancer is age.Prostate cancer is rarely diagnosed before the age of 40, then the incidence rises rapidly.PSA testing is not generally carried out on men aged under 50 unless they have significant risk factors. The rates of prostate cancer at different ages are as follows:
- 20 to 30 years: 2–8%;
- 31 to 40 years: 9–31%;
- 41 to 50 years: 3–43%;
- 51 to 60 years: 5–46%;
- 61 to 70 years: 14–70%;
- 71 to 80 years: 31–83%; and
- 81 to 90 years: 40–73%.
Genetics
Around 5–10% of prostate cancer is due to genetic defects, so men who have a family history are more at risk of developing it themselves. Men who have a first-degree relative (brother, father or son) who has had prostate cancer have a 2–3 times greater chance of developing prostate cancer themselves.This risk may be higher if the relative was diagnosed before the age of 60.
Identifying particular genes involved in prostate cancer has proved challenging. BRCA1 and BRCA2 genes are involved in a 2–5 fold increased risk of prostate cancer.
In American studies, African-American men have higher rates of developing prostate cancer than Caucasian or Hispanic men. Indigenous Australians are at a lower risk of being diagnosed with prostate cancer than non-Indigenous Australians.
Diet
The links between prostate cancer and diet has been extensively studied. Significant findings include:
- Diet high in animal fat may contribute to prostate cancer
- A diet low in vegetables may be a risk factor for prostate cancer
- Soy intake may have a modest protective effect against prostate cancer.
Other factors
- Other men who are at increased risk of prostate cancer are those who have had elevated PSA tests, or abnormal prostate biopsies in the past.
- Testosterone levels were previously thought to be related to development of prostate cancer; however, this does not appear to be the case.
- Obesity may be linked with more aggressive forms of, but not increased incidence of prostate cancer.
- Smoking has been linked to increased deaths due to prostate cancer in Caucasians.
Progression
In many cases, prostate cancer spreads so slowly that it never causes illness, and the patient will die of other causes. However, if the cancer is left untreated, it is possible that it will invade locally, spread to local pelvic lymph nodes, and eventually spread to other parts of the body (metastasise). This may eventually result in organ failure and death. Bony metastases from prostate cancer are common.
For men who choose not to undergo definitive local treatment, survival relates to the aggressiveness (histological grade) of prostate cancer.
In general, neuroendocrine carcinoma of the prostate is more aggressive and has a worse prognosis than conventional adenocarcinoma of the prostate.
Symptoms
Currently, most prostate cancer diagnosed is asymptomatic. The cancer is detected due to abnormalities in PSA levels.
In the past, prostate cancer was diagnosed due to abnormal digital rectal examination (DRE), or due to the presence of urinary symptoms such as difficulty starting urine stream, urgent rush to get to the toilet, needing to pass urine frequently at night, and dribbling of urine at the end of the stream. However, these urinary symptoms are more commonly caused by benign prostatic hyperplasia or bladder infections.
Other symptoms that may raise suspicion of prostate disease include new onset erectile dysfunction, blood in the urine, blood in the semen, or symptoms of metastasis such as bone pain.
Clinical Examination

PSA and DRE are generally performed together to check for prostate cancer. PSA testing requires a blood test to be taken.
How is it Diagnosed
A blood test is carried out to check the levels of prostate specific antigen (PSA). If a man’s PSA level is elevated above 4 ng/mL, it may indicate that he is at increased risk of prostate cancer. However, most men with elevated PSA do not have prostate cancer. It must be noted that neuroendocrine carcinoma may not be detected as accurately by PSA as adenocarcinoma of the prostate, because some forms do not secrete PSA.
If further investigation is required, the next stage is usually a prostate biopsy, which is performed using ultrasound, via the rectum. An implement passes needles into the prostate, which are withdrawn with tissue inside them. The prostate tissue is examined under a microscope to determine if cancer is present, what type it is, and how aggressive it is. Serious complications due to prostate biopsy are rare. Hospitalisation (usually for urinary infections), or urinary retention occur in less than 1% of cases.One study found the following side-effects for biopsy: blood in the semen (51%), blood in the urine (23% longer than three days), fever (3.5%) and rectal bleeding (1.3%). These symptoms were generally short term.
If cancer is identified on biopsy, the doctor will then carry out further investigations, such as removal of local lymph nodes, or CT, to determine whether the cancer has spread.
Prognosis
The risk of a man being diagnosed with any type of prostate cancer at some point in his life is 1 in 6 (17%). However, only 3% of men die from prostate cancer. At autopsy, one third of men under the age of 80 and two thirds of men over the age of 80 are found to have some form of prostate cancer.This data suggests that most men die with prostate cancer, rather than from it. At five years, 99% of men diagnosed with prostate cancer are still alive. At 10 years, 95% of men diagnosed with prostate cancer are still alive.
Determining the degree of spread of the cancer is known as ‘staging’. Cancer that is confined to the prostate gland is generally curable. Prognosis is less good if the cancer has spread locally. If the cancer has spread to other parts of the body such as the bones, it is very difficult to cure. Treatment is focused on slowing the spread of the disease, and men may still live for many years with a good quality of life. Prognosis is also related to the grade of the cancer, which in general is higher in neuroendocrine than in adenocarcinoma of the prostate. This means that neuroendocrine cancer is a more aggressive variety, so the prognosis is worse. Because it is rare, more specific figures related to prognosis are unavailable at this stage.
Treatment
- Active surveillance;
- Radical prostatectomy;
- External beam radiotherapy;
- Brachytherapy (internal radiotherapy); and
- Hormonal therapy.
Active surveillance
The risk of some small, non aggressive, localised prostate cancers ever spreading is very low. In this case the risks of treatment may outweigh the benefits. A doctor may therefore recommend ‘watchful waiting’, also known as ‘active surveillance’. The cancer and PSA levels are monitored, but no active treatment is carried out. This may be particularly appropriate for men who have a life expectancy of less than 10 or 15 years due to advanced age or other medical problems.
Treatment in Australia
If a man chooses to have treatment for the prostate cancer, the main options in Australia include surgery, radiotherapy and hormone therapy. There is no good quality evidence that one approach is superior to the others, so treatment choice is based on the following considerations:
- Age, general medical condition and other medical problems. If a man has a less than 10 year life expectancy, definitive treatment may not be recommended.
- Serum PSA and Gleason Score. The Gleason Score is determined following prostate biopsy and is a measure of the aggressiveness of the cancer. If these levels are high, it may indicate the cancer is more aggressive, so more aggressive treatment may be justified.
- Spread of the prostate cancer. If the cancer has already spread to bones and other organs, treatment aiming to cure the disease is not likely to be effective. Treatment options such as palliative radiotherapy may then be used to treat pain and other symptoms.
- Outcomes associated with treatment options. Various treatments may have different outcomes, and need to be considered in the context of the individual patient, and the specifics (location, aggressiveness etc) of their disease. There is not a single treatment that results in the best outcome for all patients with prostate cancer.
- Side effects and quality of life implications related to treatment options. Each of the treatment options has different side effects. Each patient needs to evaluate how he feels about the possibility of such side effects (such as sexual dysfunction), and weigh up the risks and benefits of each procedure to determine which is most appropriate for him.
Radical prostatectomy
Radiotherapy (external beam radiation therapy)
External beam radiation therapy (EBRT) may be used with RP or hormonal therapy to treat prostate cancer. Beams of radiotherapy (like x-rays) are targeted at the prostate by a machine that moves around the body. The rays kill the cancer cells. Small tattoos may be placed on the skin to make sure the beam is in exactly the right place, and minimise healthy tissue being damaged. This procedure is performed in an outpatient setting, and men are usually able to maintain their normal activities. Radiotherapy may lead to sexual dysfunction in 20-45% of cases, urinary incontinence in 2-16%, and bowel dysfunction in 6-25% of men who were previously normal in these areas.
Brachytherapy (internal radiotherapy)
There are two main types of brachytherapy. One involves the insertion of a radioactive pellet into the prostate (seeding). The pellet loses its radioactivity over time, and is not removed. This option may be used for low Gleason score, low PSA, small localised prostate cancer. Side effects such as painful urination, poor urine flow and bladder irritation are common. These can start a month after treatment and gradually get better over 12 months. The second type of brachytherapy involves insertion of wide bore needles into the prostate, through which radioactive sources can be administered. After several days of treatment, the needles are removed. This type of brachytherapy is used in conjunction with EBRT, for larger more aggressive tumours. The needles may cause local discomfort, usually relieved by painkillers. Other side effects are similar to those for EBRT.
Hormonal therapy (androgen deprivation therapy)

Side effects of ADT include hot flushes, loss of libido (sex drive), weight gain, erectile dysfunction (impotence), gynecomastia (swelling of the breasts), fluid retention, softening of the bones (osteoporosis) and mood swings.
Palliative treatments
In some cases, when prostate cancer is diagnosed, it is too advanced to be cured by surgery or radiotherapy. In these cases, treatment is referred to as ‘palliative’. Palliative treatment is designed to relieve symptoms, maintain quality of life, and in many cases, prolong life but it will not cure the disease.
- Transuretheral Resection of the Prostate (TURP) may be carried out to relieve obstructive symptoms due to the cancer.
- Hormone therapy- ADT is a mainstay of treatment for cancer that is not resectable. Eventually, the cancer will no longer respond to ADT. At this point as prognosis is poor, with an average survival of 1 year once symptoms occur.
- Bisphosphonates and analgesia may be used to treat bone metastases
- EBRT may be used to treat pain related to bone metastases, and improves pain in 80-90% of cases with complete pain response in 50-60% of cases.
Relative side effects of different treatments
Radical prostatectomy and brachytherapy result in higher rates of urinary incontinence and sexual dysfunction than EBRT. Bilateral nerve sparing surgery (not possible in all cases) diminishes this difference.
- RT causes more bowel dysfunction than surgery.
- Brachytherapy causes more obstructive and irritative urinary symptoms.
Treatment recommendations
Good quality evidence related to the best approach to prostate cancer is limited, however the following treatment strategies have been suggested:
- Low-risk disease: Radical prostatectomy, brachytherapy, or EBRT.
- Intermediate-risk disease: EBRT or surgery. The role of brachytherapy alone is controversial. Brachytherapy plus EBRT may be superior to EBRT alone, though this is controversial.
- High-risk disease: Radical prostatectomy on its own or followed by EBRT, or EBRT plus ADT. EBRT or brachytherapy alone are not generally appropriate.
More information
 |
For more information on prostate cancer, including diagnoses, types of treatments, and some useful tools, videos and animations, see Prostate Cancer. |
References
- Epstein JI, Yang XJ. Prostate Biopsy Interpretation (3rd edition). Philadelphia: Lippincott Williams and Wilkins; 2002. [Book]
- di Sant’Agnese PA. Neuroendocrine differentiation in carcinoma of the prostate. Diagnostic, prognostic, and therapeutic implications. Cancer. 1992;70(1 Suppl):254-68. [Abstract]
- Braunwald E, Fauci AS, Kasper DL, et al. Harrison’s Principles of Internal Medicine (15th edition). New York: McGraw-Hill Publishing; 2001. [Book]
- Montironi R. Prognostic factors in prostate cancer. BMJ. 2001;322(7283):378-9. [Abstract | Full text]
- Reyes A, Moran CA. Low-grade neuroendocrine carcinoma (carcinoid tumor) of the prostate. Arch Pathol Lab Med. 2004;128(12):e166-8. [Abstract | Full text]
- Cohen RJ, Shannon BA, Phillips M, et al. Central zone carcinoma of the prostate gland: a distinct tumor type with poor prognostic features. J Urol. 2008;179(5):1762-7. [Abstract]
- Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008;58(2):71-96. [Abstract | Full text]
- Position statement: Prostate cancer screening [online]. Sydney, NSW: Cancer Council Australia; June 2010 [cited 18 June 2011]. Available from: URL link
- Roder D. Epidemiology of cancer in Indigenous Australians: Implications for service delivery. Paper presented at the 9th National Rural Health Conference: Standing up for rural health: Learning from the past, action in the future. Albury, NSW: 7-10 March 2007. Available from: URL link
- Ries LAG, Eisner MP, Kosary CL, et al (eds). SEER Cancer Statistics Review, 1973-1999. Bethesda, MD: National Cancer Institute; 2002. Available from: URL link
- Potosky AL, Miller BA, Albertsen PC, Kramer BS. The role of increasing detection in the rising incidence of prostate cancer. JAMA. 1995;273(7):548-52. [Abstract]
- Mettlin C, Jones G, Averette H, et al. Defining and updating the American Cancer Society guidelines for the cancer-related checkup: Prostate and endometrial cancers. CA Cancer J Clin. 1993;43(1):42-6. [Abstract]
- American Urological Association. Early detection of prostate cancer and use of transrectal ultrasound. In: American Urological Association 1992 Policy Statement Book. Baltimore, MD: Williams & Wilkins; 1992.
- Bouchardy C, Fioretta G, Rapiti E, et al. Recent trends in prostate cancer mortality show a continuous decrease in several countries. Int J Cancer. 2008;123(2):421-9. [Abstract | Full text]
- Ries LAG, Melbert D, Krapcho M, et al (eds). SEER Cancer Statistics Review, 1975-2004. Bethesda, mD: National Cancer Institute; 2007. Available from: URL link
- Whitmore WF Jr. Natural history of low-stage prostatic cancer and the impact of early detection. Urol Clin North Am. 1990;17(4):689-97. [Abstract]
- Mettlin, C, Lee, F, Drago, J, Murphy, GP. The American Cancer Society National Prostate Cancer Detection Project. Findings on the detection of early prostate cancer in 2425 men. Cancer 1991; 67:2949.
- Johns LE, Houlston RS 2003. A systematic review and meta-analysis of familial prostate cancer risk. BJU Int 91(9):789-94.
- Ries, LA, Eisner, MP, Kosary, CL, et al, eds. SEER Cancer Statistics Review, 1973-1999 National Cancer Institute [online]. 2002 [cited 2008 October 30]. Available from: URL: http://seer.cancer.gov/csr/1973_1999/
- Hankey, BF, Feuer, EJ, Limin, X, et al. Cancer surveillance series: Interpreting trends in prostate cancer – Part 1: Evidence of the effects of screening in recent prostate cancer incidence, mortality, and survival rates. J NatlCancer Inst 1999; 91:1017.
- Delongchamps, NB, Singh, A, Haas, GP. The role of prevalence in the diagnosis of prostate cancer. Cancer Control 2006; 13:158.
- Stamey, TA. Making the most out of six systematic sextant biopsies. Urology 1995; 45:2.
- Thompson, D, Easton, DF. Cancer Incidence in BRCA1 Mutation Carriers. J Natl Cancer Inst 2002; 94:1358.
- Struewing, JP, Hartge, P, Wacholder, S, et al. The risk of cancer associated with specific mutations of BRCA1 and BRCA2 among Ashkenazi Jews. N Engl J Med 1997; 336:1401.
- Cancer risks in BRCA2 mutation carriers.The Breast Cancer Linkage Consortium. J Natl Cancer Inst 1999; 91:1310.
- Edwards, SM, Kote-Jarai, Z, Meitz, J, et al. Two Percent of Men with Early-Onset Prostate Cancer Harbor Germline Mutations in the BRCA2 Gene. Am J Hum Genet 2003; 72:1.
- Risch, HA, McLaughlin JR, Cole DE, Rosen B, Bradley L, Kwan E, et al. Population. J Natl Cancer Inst 2006; 98:1694.
- Liede, A, Karlan, BY, Narod, SA. Cancer risks for male carriers of germline mutations in BRCA1 or BRCA2: a review of the literature. J Clin Oncol 2004; 22:735.
- Roddam, AW, Allen, NE, Appleby, P, Key, TJ. Endogenous sex hormones and prostate cancer: a collaborative analysis of 18 prospective studies. J Natl Cancer Inst 2008; 100:170.
- Huxley R. Asia Pacific Cohort Studies Collaboration. The impact of modifiable risk factors on mortality from prostate cancer in populations of the Asia-Pacific region. Asian Pacific Journal of Cancer Prevention: Apjcp. 8(2):199-205, 2007 Apr-Jun.
- Andersson, SO, Wolk, A, Bergstrom, R, et al. Body size and prostate cancer: a 20-year follow-up study among 135006 Swedish construction workers. J Natl Cancer Inst 1997; 89:385.
- Calle, EE, Rodriguez, C, Walker-Thurmond, K, et al. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of U.S. adults. N Engl J Med 2003; 348:1625.
- Amling, CL, Riffenburgh, RH, Sun, L, et al. Pathologic variables and recurrence rates as related to obesity and race in men with prostate cancer undergoing radical prostatectomy. J Clin Oncol 2004; 22:439.
- Wright, ME, Chang, SC, Schatzkin, A, et al. Prospective study of adiposity and weight change in relation to prostate cancer incidence and mortality. Cancer 2007; 109:675.
- Schulman, CC, Ekane, S, Zlotta, AR. Nutrition and prostate cancer: evidence or suspicion?. Urology 2001; 58:318.
- Chan, JM, Gann, PH, Giovannucci, EL. Role of diet in prostate cancer development and progression. J Clin Oncol 2005; 23:8152.
- Kolonel, LN, Nomura, AM, Cooney, RV. Dietary fat and prostate cancer: Current status. J Natl Cancer Inst 1999; 91:414.
- Colli, JL, Colli, A. International comparisons of prostate cancer mortality rates with dietary practices and sunlight levels. Urol Oncol 2006; 24:184.
- Giovannucci, E, Rimm, EB, Colditz, GA, et al. A prospective study of dietary fat and risk of prostate cancer. J Natl Cancer Inst 1993; 85:1571.
- Cohen, JH, Kristal, AR, Stanford, JL. Fruit and vegetable intakes and prostate cancer risk. J Natl Cancer Inst 2000; 92:61.
- Jian, L, Du, CJ, Lee, AH, Binns, CW. Do dietary lycopene and other carotenoids protect against prostate cancer?. Int J Cancer 2005; 113:1010.
- Kolonel, LN, Hankin, JH, Whittemore, AS, et al. Vegetables, fruits, legumes and prostate cancer: a multiethnic case-control study. Cancer Epidemiol Biomarkers Prev 2000; 9:795.
- Jacobsen, BK, Knutsen, SF, Fraser, GE. Does high soy milk intake reduce prostate cancer incidence? The Adventist Health Study (United States). Cancer Causes Control 1998; 9:553.
- Yan, L, Spitznagel, EL. Meta-analysis of soy food and risk of prostate cancer in men. Int J Cancer 2005; 117:667.
- Catalona, WJ, Richie, JP, Ahmann, FR, et al. Comparison of digital rectal examination and serum prostate specific antigen in the early detection of prostate cancer: results of a multicenter clinical trial of 6,630 men. J Urol 1994; 151:1283.
- Pedersen, KV, Carlsson, P, Varenhorst, E, Lofman, O. Screening for carcinoma of the prostate by digital rectal examination in a randomly selected population. BMJ 1990; 300:1041.
- Chodak, GW, Keller, P, Schoenberg, HW. Assessment of screening for prostate cancer using the digital rectal examination. J Urol 1989; 141:1
- Gustafsson, O, Norming, U, Almgard, LE, et al. Diagnostic methods in the detection of prostate cancer: A study of a randomly selected population of 2,400 men. J Urol 1992; 148:1827.
- Yamamoto, T, Ito, K, Ohi, M, Kubota, Y. Diagnostic significance of digital rectal examination and transrectal ultrasonography in men with prostate-specific antigen levels of 4 NG/ML or less. Urology 2001; 58:994.
- Capella C, Usellini L, Buffa R, Frigerio B, Solcia E. The endocrine component of prostatic carcinomas, mixed adenocarcinoma-carcinoid tumours and nontumour prostate: histochemical and ultrastructural identification of the endocrine cells. Histopathology 1981;5:175-192.
- di Sant’Agnese PA. Neuroendocrine cells of the prostate and neuroendocrine differentiation in prostatic carcinoma: a review of morphologic aspects. Urology 1998;51:121-124.
- Azumi N, Shibuya H, Ishikura M. Primary prostatic carcinoid tumor with intracytoplasmic prostatic acid phosphatase and prostate-specific antigen. Am J Surg Pathol 1984;8:545-550.
- Ghali VS, Garcia RL. Prostatic adenocarcinoma with carcinoidal features producing adrenocorticotropic syndrome: immunohistochemical study and review of the literature. Cancer 1984;15:1043-1048.
- Slater D. Carcinoid tumor of the prostate associated with inappropriate ACTH secretion. Br J Urol 1985;57:591-592.
- Turbat-Herrera EA, Herrera GA, Gore I, Lott RL, Grizzle WE, Bonnin JM. Neuroendocrine differentiation in prostatic carcinomas: a retrospective autopsy study. Arch Pathol Lab Med 1988;112:1100-1105.
- Varma, M, Lee, MW, Tamboli, P, et al. Morphologic criteria for the diagnosis of prostatic adenocarcinoma in needle biopsy specimens. A study of 250 consecutive cases in a routine surgical pathology practice. Arch Pathol Lab Med 2002; 126:554.
- Johansson, J, Andrén, O, Andersson, S et al. Natural History of Early, Localized Prostate Cancer. JAMA. 2004;291:2713-2719.
- Chodak, GW, Thisted, RA, Gerber, GS, et al. Results of conservative management of clinically localized prostate cancer. N Engl J Med 1994; 330:242.
- Brenner, H, Arndt, V. Long-term survival rates of patients with prostate cancer in the prostate-specific antigen screening era: population-based estimates for the year 2000 by period analysis. J Clin Oncol 2005; 23:441.
- Cooperberg, MR, Moul, JW, Carroll, PR. The changing face of prostate cancer. J Clin Oncol 2005; 23:8146.
- Miller, DC, Hafez, KS, Stewart, A, et al. Prostate carcinoma presentation, diagnosis, and staging: an update form the National Cancer Data Base. Cancer 2003; 98:1169.
- Crook, J, Lukka, H, Klotz, L, Bestic, N. Systematic overview of the evidence for brachytherapy in clinically localized prostate cancer. CMAJ 2001; 164:975.
- Frydenberg M. Wijesinha S. Diagnosing prostate cancer – what GPs need to know. Australian Family Physician. 36(5):345-7, 2007 May.
- Catalona, WJ, Richie, JP, Ahmann, FR, et al. Comparison of digital rectal examination and serum prostate specific antigen in the early detection of prostate cancer: results of a multicenter clinical trial of 6,630 men. J Urol 1994; 151:1283.
- Pedersen, KV, Carlsson, P, Varenhorst, E, Lofman, O. Screening for carcinoma of the prostate by digital rectal examination in a randomly selected population. BMJ 1990; 300:1041.
- Chodak, GW, Keller, P, Schoenberg, HW. Assessment of screening for prostate cancer using the digital rectal examination. J Urol 1989; 141:1136.
- Gustafsson, O, Norming, U, Almgard, LE, et al. Diagnostic methods in the detection of prostate cancer: A study of a randomly selected population of 2,400 men. J Urol 1992; 148:1827.
- Yamamoto, T, Ito, K, Ohi, M, Kubota, Y. Diagnostic significance of digital rectal examination and transrectal ultrasonography in men with prostate-specific antigen levels of 4 NG/ML or less. Urology 2001; 58:994.
- Richie, JP, Catalona, WJ, Ahmann, FR, et al. Effect of patient age on early detection of prostate cancer with serum prostate-specific antigen and digital rectal examination. Urology 1993; 42:365.
- Crawford, ED, Schutz, MJ, Clejan, S, et al. The effect of digital rectal examination on prostate-specific antigen levels. JAMA 1992; 267:2227.
- Carroll, P, Coley, C, McLeod, D, et al. Prostate specific antigen best practice policy part I: early detection and diagnosis of prostate cancer. Urology 2001; 57:217.
- Smith, RA, von Eschenbach, AC, Wender, R, et al. American Cancer Society guidelines for the early detection of cancer: update of early detection guidelines for prostate, colorectal, and endometrial cancers. Also: update 2001-testing for early lung cancer detection. CA Cancer J Clin 2001; 51:38.
- Chong CC. Austen L. Kneebone A. Lalak A. Jalaludin B. Patterns of practice in the management of prostate cancer: results from multidisciplinary surveys of clinicians in Australia and New Zealand in 1995 and 2000. BJU International. 97(5):975-80, 2006 May.
- Dorr, VJ, Williamson, SK, Stephens, RL. An evaluation of prostate-specific antigen as a screening test for prostate cancer. Arch Intern Med 1993; 153:2529.
- Harris R & Lohr KN 2002. Screening for prostate cancer: an update of the evidence for the US Preventive Services Task Force. Ann Intern Med 137(11):917-29.
- Abdalla, I, Basu, A, Hellman, S. An evidence-based analysis of the management of localized prostate cancer. Cancer J 2002; 8:40.
- Thompson, I, Thrasher, JB, Aus, G, et al. Guideline for the management of clinically localized prostate cancer: 2007 update. J Urol 2007; 177:2106.
- Coley, CM, Barry, MJ, Fleming, C, et al. Early detection of prostate cancer. Part I: prior probability and effectiveness of tests. Ann Intern Med 1997; 126:394.
- Partin, AW, Carter, HB, Chan, DW, et al. Prostate specific antigen in the staging of localized prostate cancer: influence of tumor differentiation, tumor volume and benign hyperplasia. J Urol 1990; 143:747.
- Lange, PH, Ercole, CJ, Lightner, DJ, et al. The value of serum prostate specific antigen determinations before and after radical prostatectomy. J Urol 1989; 141:873.
- Hudson, MA, Bahnson, RR, Catalona, WJ. Clinical use of prostate specific antigen in patients with prostate cancer. J Urol 1989; 142:1011.
- Thompson, IM, Goodman, PJ, Tangen, CM, et al. The influence of finasteride on the development of prostate cancer. N Engl J Med 2003; 349:215.
- Thompson, IM, Pauler, DK, Goodman, Pj, et al. Prevalence of prostate cancer among men with a prostate-specific antigen level<4.0 ng/mL. N Engl J Med 2004; 350:2239.
- Catalona, WJ, Smith, DS, Ornstein, DK. Prostate cancer detection in men with serum PSA concentrations of 2.6 to 4.0 ng/mL and benign prostate examination. JAMA 1997; 277:1452.
- Makarov, DV, Humphreys, EB, Mangold, LA, et al. Pathological outcomes and biochemical progression in men with T1c prostate cancer undergoing radical prostatectomy with prostate specific antigen 2.6 to 4.0 vs 4.1 to 6.0 ng/ml. J Urol 2006; 176:554.
- National Comprehensive Cancer Network (NCCN) guidelines available online at www.nccn.org/professionals/physician_gls/default.asp (Accessed October 2, 2008)
- Carroll, P, Coley, C, McLeod, D, et al. Prostate-specific antigen best practice policy-part I: early detection and diagnosis of prostate cancer. Urology 2001; 57:217.
- Polascik, TJ, Oesterling, JE, Partin, AW. Prostate specific antigen: a decade of discovery-what we have learned and where we are going. J Urol 1999; 162:293.
- Eastham, JA, Riedel, E, Scardino, PT, et al. Variation of serum prostate-specific antigen levels: an evaluation of year to year fluctuations. JAMA 2003; 289:2695.
- Obek, C, Ozkan, B, Tunc, B, et al. Comparison of 3 different methods of anesthesia before transrectal prostate biopsy: a prospective randomized trial. J Urol 2004; 172:502.
- Yun, TJ, Lee, HJ, Kim, SH, et al. Does the intrarectal instillation of lidocaine gel before periprostatic neurovascular bundle block during transrectal ultrasound guided prostate biopsies improve analgesic efficacy? A prospective, randomized trial. J Urol 2007; 178:103.
- Holmberg, L, Bill-Axelson, A, Helgesen, F, et al. A randomized trial comparing radical prostatectomy with watchful waiting in early prostate cancer. N Engl J Med 2002; 347:781.
- Bill-Axelson, A, Holmberg, L, Ruutu, M, et al. Radical prostatectomy versus watchful waiting in early prostate cancer. N Engl J Med 2005; 352:1977.
- Wilt, TJ, Brawer, MK. The Prostate Cancer Intervention Versus Observation Trial: a randomized trial comparing radical prostatectomy versus expectant management for the treatment of clinically localized prostate cancer. J Urol 1994; 152:1910.
- Dahm P. Kunz R. Schunemann H. Evidence-based clinical practice guidelines for prostate cancer: the need for a unified approach. Current Opinion in Urology. 17(3):200-7, 2007 May.
- Haas, GP, Delongchamps, NB, Jones, RF, et al. Needle biopsies on autopsy prostates: sensitivity of cancer detection based on true prevalence. J Natl Cancer Inst 2007; 99:1484.
- Epstein, JI, Walsh. PC, Carmichael, M, Brendler, CM. Pathologic and clinical findings to predict tumor extent of nonpalpable (stage T1c) prostate cancer. JAMA 1994; 271:368.
- Albertsen, PC, Hanley, JA, Penson, DF, et al. 13-year outcomes following treatment for clinically localized prostate cancer in a population based cohort. J Urol 2007; 177:932.
- Cooperberg, MR, Lubeck, DP, Meng, MV, et al. The changing face of low-risk prostate cancer: trends in clinical presentation and primary management. J Clin Oncol 2004; 22:2141.
- Brenner, H, Arndt, V. Long-term survival rates of patients with prostate cancer in the prostate-specific antigen screening era: population-based estimates for the year 2000 by period analysis. J Clin Oncol 2005; 23:441.
- Walz, J, Gallina, A, Saad, F, et al. A nomogram predicting 10-year life expectancy in candidates for radical prostatectomy or radiotherapy for prostate cancer. J Clin Oncol 2007; 25:3576.
- Wei, JT, Dunn, RL, Sandler, HM, et al. Comprehensive comparison of health-related quality of life after contemporary therapies for localized prostate cancer. J Clin Oncol 2002; 20:557.
- Miller, DC, Sanda, MG, Dunn, RL, et al. Long-term outcomes among localized prostate cancer survivors: health-related quality of life changes after radical prostatectomy, external radiation, and brachytherapy. J Clin Oncol 2005; 23:2772.
- Ung, JO, Richie, JP, Chen, MH, et al. Evolution of the presentation and pathologic and biochemical outcomes after radical prostatectomy for patients with clinically localized prostate cancer diagnosed during the PSA era. Urology 2002; 60:458.
- Begg, CB, Riedel, ER, Bach, PB, et al. Variations in morbidity after radical prostatectomy. N Engl J Med 2002; 346:1138.
- Lu-Yao, GL, Albertsen, P, Warren, J, Yao, SL. Effect of age and surgical approach on complications and short-term mortality after radical prostatectomy- a population based study. Urology 1999; 54:301.
- Stanford, JL, Feng, Z, Hamilton, AS, et al. Urinary and sexual function after radical prostatectomy for clinically localized prostate cancer: the Prostate Cancer Outcomes Study. JAMA 2000; 283:354.
- Hamilton, AS, Stanford, JL, Gilliland, FD, et al. Health outcomes after external-beam radiation therapy for clinically localized prostate cancer: results from the Prostate Cancer Outcomes Study. J Clin Oncol 2001; 19:2517.
- Crook, J, Lukka, H, Klotz, L, Bestic, N. Systematic overview of the evidence for brachytherapy in clinically localized prostate cancer. CMAJ 2001; 164:975.
- Crawford, ED, Eisenberger, MA, McLeod, DG, et al. A controlled trial of leuprolide with and without flutamide in prostatic carcinoma. N Engl J Med 1989; 321:419.
- Denis, LJ, Keuppens, F, Smith, PH, et al. Maximal androgen blockade: final analysis of EORTC phase III trial 30853. EORTC Genito-Urinary Tract Cancer Cooperative Group and the EORTC Data Center. Eur Urol 1998; 33:144.
- Janknegt, RA, Abbou, CC, Bartoletti, R, et al. Orchiectomy and nilutamide or placebo as treatment of metastatic prostatic cancer in a multinational double-blind randomized trial. J Urol 1993; 149:77.
- Dijkman, GA, Janknegt, RA, De Reijke, TM, Debruyne, FM. Long-term efficacy and safety of nilutamide plus castration in advanced prostate cancer, and the significance of early prostate specific antigen normalization. International Anandron Study Group. J Urol 1997; 158:160.
- Eisenberger, MA, Blumenstein, BA, Crawford, ED, et al. Bilateral orchiectomy with or without flutamide for metastatic prostate cancer. N Engl J Med 1998; 339:1036.
- Varghese S, Grossfeld G. The prostate gland: malignancies other than adenocarcinomas. Radiologic Clinics of North America – Volume 38 (1). 2000.
- Efros MD, et al: Unusual primary prostatic malignancies. Urology 39:407, 1992.
- Matzkin H, Braf Z: Paraneoplastic syndromes associated with prostatic carcinoma. J Urol 138:1129, 1987.
- Tetu B, et al: Small cell carcinoma of prostate associated with myasthenic (Eaton-Lambert) syndrome. Urology 33:148, 1989.
- Australian Institute of Health and Welfare & Australasian Association of Cancer Registries. Cancer in Australia: an overview, 2010 Cat. no. CAN 56 [online]. Canberra: AIHW 2010. [cited June 18 2011]. Available from: URL Link
- Roder D, Currow D. Cancer in Aboriginal and Torres Strait Islander People of Australia. Asian Pacific J Cancer Pre., 2008; (10):729-733. [Abstract ?| Full Text]
All content and media on the HealthEngine Blog is created and published online for informational purposes only. It is not intended to be a substitute for professional medical advice and should not be relied on as health or personal advice. Always seek the guidance of your doctor or other qualified health professional with any questions you may have regarding your health or a medical condition. Never disregard the advice of a medical professional, or delay in seeking it because of something you have read on this Website. If you think you may have a medical emergency, call your doctor, go to the nearest hospital emergency department, or call the emergency services immediately.

 Radical prostatectomy and brachytherapy result in higher rates of urinary incontinence and sexual dysfunction than EBRT. Bilateral nerve sparing surgery (not possible in all cases) diminishes this difference.
Radical prostatectomy and brachytherapy result in higher rates of urinary incontinence and sexual dysfunction than EBRT. Bilateral nerve sparing surgery (not possible in all cases) diminishes this difference.





