- What is Gastrointestinal Stromal Tumours (GISTs)?
- Statistics
- Risk Factors
- Progression
- Symptoms
- Clinical Examination
- How is it Diagnosed
- Prognosis
- Treatment
- References
What is Gastrointestinal Stromal Tumours (GISTs)?
Statistics
Due to the lack of a complete definition and classification the precise incidence of GIST is unknown, however, it is estimated to be roughly 10-20 per million people per year. GIST accounts for less than 1% of all gastrointestinal cancers. More than 90% of GISTs occur in adults over 40 years of age.
GISTs are equally as common in males as females and do not appear to be associated with geographic location, ethnicity, race or occupation.
Risk Factors
Currently, there are no known specific risk factors for developing GIST.
Most cases occur are not inherited (sporadic). However, there are reports of several families in which multiple family members are affected. In these families the mutation is passed down from parent to child and occurs in every cell in their body not just the cancerous cells (as in sporadic cases). Hereditary syndromes in which GISTs occur include:
- Familial gastrointestinal stromal tumour syndrome (FGS): Appears to be an autosomal dominant syndrome equally affecting males and females. Typically individuals are diagnosed at a younger age than for sporadic cases and have multiple GISTs. Members may also show skin changes including hyperpigmentation, or increased numbers of nevi.
- Carney triad: Triad of GIST, pulmonary chondromas and extra-adrenal paragangliomas first described in 1977. Most reported cases have been in females aged less than 30 years. Family history is not common and it is thought to be a sporadic condition.
- Carney-Stratakis syndrome: Autosomal dominant predisposition syndrome in which individuals develop either paraganglioma, GIST or both.
- Neurofibromatosis type 1: Complex disorder affecting 1 in every 3,000 live births. As in FGS, tumours tend to be multiple, are most common in the small bowel and diagnosed earlier than sporadic cases.
Symptoms
GISTs present with signs and symptoms related to the presence of a mass or bleeding. Bleeding is the most common symptom which may occur into the abdominal cavity resulting in acute abdominal pain or into the gastrointestinal tract lumen which may present as vomiting blood (haematemesis), blood stained stools (melena) or anaemia. Abdominal masses are less common and depending on their location can cause a variety of symptoms including obstruction.
Many people present with vague symptoms, which are not disease specific, including nausea, vomiting, abdominal discomfort, weight loss or early satiety. As a result, up to 50% of GISTs will have already metastasised by the time they are diagnosed.
Only 70% of individuals are symptomatic, 20% do not have any symptoms and 10% have a GIST detected at autopsy.
Clinical Examination
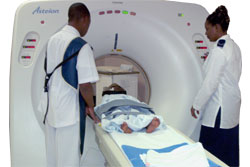
Typically it will involve imaging studies including x-ray, computed tomography (CT), and/or magnetic resonance imaging (MRI).
Once a lesion has been identified, a pathologist will need to confirm whether the lesion is cancerous or not and if so what type of tumour it represents
How is it Diagnosed
A diagnosis is made based on the location and tissue type of the identified mass. Molecular studies can be used to identify GISTs with specific mutations, which may make them either more or less responsive to treatment.
Prognosis
GISTs exhibit an uncertain clinical behaviour. They vary from benign to malignant.
Several factors including size, appearance when examined microscopically, cellular characteristics and their genetics have been examined over many years, although, we are not yet unable to predict their behaviour.
Treatment
Depending on the stage of the disease, several doctors may be involved in the management including an oncologist (doctor specialising in cancers), gastroenterologist (doctor specialising in the gastrointestinal tract) and/or a surgeon.
There are several questions that you may wish to ask your doctor including:
- The location of the GIST
- If the GIST has spread to other regions
- How it is best treated
- If there any other treatments available
- The pros and cons of any treatments offered
- The side effects of treatments offered
- How treatment side effects will be managed
- The likely success of treatment
Local disease

If the tumour is large and its complete removal would lead to significant morbidity or loss of organ function, a medication called imatinib (Glivec) may be administered prior to its removal in an attempt to reduce its size and optimise the outcome. Partial removal is generally only performed in palliative (end of life) cases where you are not aiming for cure, but rather trying to improve quality of life by minimising symptoms.
Metastatic or recurrent disease
For recurrent disease or disease that has spread elsewhere (metastatic) first line treatment involves the administration of imatinib with or without surgery. In scientific studies 50% of individuals with GIST responded to imatinib and approximately 75-85% showed at least disease stabilisation. Two-year survival rates after imatinib therapy are approximately 70% and 50% of individuals will not show disease progression.
Current data shows a dose of 400mg/day is safe, efficacious and well tolerated. Increasing doses may be considered in cases of disease progression, resistance or for individuals with specific genetic characteristics. Side effects of imatinib include oedema (swelling), nausea, muscle cramps, diarrhoea, headache, dermatitis, fatigue, vitiligo (skin pigmentation), hypothyroidism and abdominal pain. Discontinuation of treatment is associated with a high risk of relapse and for this reason, treatment should only be discontinued in cases where the disease continues to progress despite treatment, unacceptable side effects or the individual refuses further treatment.
Some GISTs do not respond to imatinib and in these cases alternative medications may be used. Sunitinib (Sutent) has been approved for use in GISTs not responsive to imatinib. This drug works in a similar way but seems to be more efficacious in some of the tumours poorly responsive to imatinib. Other agents are currently being investigated in scientific studies.
GISTs are poorly responsive to conventional chemotherapy (<10% response) and the use of radiotherapy is restricted due to resulting toxicity to surrounding structures.
Follow-up
As GISTs have an unpredictable behaviour, long term follow up is essential. This should involve regular physical examination and imaging studies. CT imaging is recommended 3-6 monthly for at least 5 years. Other investigations including endoscopy may also be used for follow up.
More information
 |
For more information on stomach cancer, including different types of cancer affecting the stomach, see Stomach Cancer. |
References
- Stamatakos M, Douzinas E, Stefanaki C, et al. Gastrointestinal stromal tumor. World J Surg Oncol. 2009;7:61. [Abstract | Full text]
- Agarwal R, Robson M. Inherited predisposition to gastrointestinal stromal tumor. Hematol Oncol Clin North Am. 2009;23(1):1-13. [Abstract]
- Nishida T, Hirota S, Taniguchi M, et al. Familial gastrointestinal stromal tumours with germline mutation of the KIT gene. Nat Genet. 1998;19(4):323-4. [Abstract]
- Carney JA, Sheps SG, Go VL, Gordon H. The triad of gastric leiomyosarcoma, functioning extra-adrenal paraganglioma and pulmonary chondroma. N Engl J Med. 1977;296(26):1517-8. [Abstract]
- McWhinney SR, Pasini B, Stratakis CA. Familial gastrointestinal stromal tumors and germ-line mutations. N Engl J Med. 2007;357(10):1054-6. [Full text]
- Takazawa Y, Sakurai S, Sakuma Y, et al. Gastrointestinal stromal tumors of neurofibromatosis type I (von Recklinghausen’s disease). Am J Surg Pathol. 2005;29(6):755-63. [Abstract]
- Kinoshita K, Hirota S, Isozaki K, et al. Absence of c-kit gene mutations in gastrointestinal stromal tumours from neurofibromatosis type 1 patients. J Pathol. 2004;202(1):80-5. [Abstract]
- Rubin BP, Antonescu CR, Scott-Browne JP, et al. A knock-in mouse model of gastrointestinal stromal tumor harboring kit K641E. Cancer Res. 2005;65(15):6631-9. [Abstract | Full text]
- Graadt van Roggen JF, van Velthuysen ML, Hogendoorn PC. The histopathological differential diagnosis of gastrointestinal stromal tumours. J Clin Pathol. 2001;54(2):96-102. [Abstract | Full text]
- Akahoshi K, Sumida Y, Matsui N, et al. Preoperative diagnosis of gastrointestinal stromal tumor by endoscopic ultrasound-guided fine needle aspiration. World J Gastroenterol. 2007;13(14):2077-82. [Abstract | Full text]
- Tornillo L, Terracciano LM. An update on molecular genetics of gastrointestinal stromal tumours. J Clin Pathol. 2006;59(6):557-63. [Abstract | Full text]
- Corless CL, Fletcher JA, Heinrich MC. Biology of gastrointestinal stromal tumors. J Clin Oncol. 2004;22(18):3813-25. [Abstract | Full text]
- Hoeben A, Schöffski P, Debiec-Rychter M. Clinical implications of mutational analysis in gastrointestinal stromal tumours. Br J Cancer. 2008;98(4):684-8. [Abstract | Full text]
- Blackstein ME, Blay JY, Corless C, et al. Gastrointestinal stromal tumours: Consensus statement on diagnosis and treatment. Can J Gastroenterol. 2006;20(3):157-63. [Abstract | Full text]
- Parfitt JR, Streutker CJ, Riddell RH, Driman DK. Gastrointestinal stromal tumors: A contemporary review. Pathol Res Pract. 2006;202(12):837-47. [Abstract]
- Joensuu H. Gastrointestinal stromal tumor (GIST). Ann Oncol. 2006;17(Suppl 10):x280-6. [Abstract | Full text]
All content and media on the HealthEngine Blog is created and published online for informational purposes only. It is not intended to be a substitute for professional medical advice and should not be relied on as health or personal advice. Always seek the guidance of your doctor or other qualified health professional with any questions you may have regarding your health or a medical condition. Never disregard the advice of a medical professional, or delay in seeking it because of something you have read on this Website. If you think you may have a medical emergency, call your doctor, go to the nearest hospital emergency department, or call the emergency services immediately.







