- What is growth hormone?
- What are the actions of growth hormone?
- Why is growth hormone used to manage obesity?
- How effective is growth hormone in the management of obesity?
- What are the risks of growth hormone use?
What is a hormone?
Hormones are chemicals that are released by cells in the body and are detected by other cells, altering the way they function. They act as ‘chemical messengers’, and play a large role in how the body operates. ‘Artificial’ hormones may be created or synthesised and administered in the form of drugs to treat medical conditions.
What is growth hormone?
Growth hormone (GH) is a hormone produced by the cells of the pituitary gland, a small gland found in the brain. GH production begins early in the womb and continues throughout life, though at a lesser rate. GH release is altered by several factors.
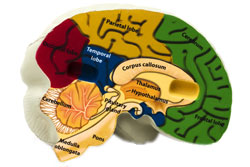
- Growth hormone releasing hormone (GHRH), a hormone released by the hypothalamus (a part of the brain);
- Ghrelin, a hormone produced in the gut;
- Fasting;
- High protein meals;
- Low blood sugar levels (hypoglycaemia). Insulin can be used to create low blood sugar levels. In normal circumstances, this would result in increased GH secretion. If this does not occur, GH deficiency may be diagnosed.
- The main female sex hormone oestrogen;
- Other hormones such as dopamine, alpha-adrenergic agonists (released during times of stress) and beta-adrenergic antagonists (beta blockers); and
- Exercise, infection, and trauma.
GH secretion is reduced by:
- Somatostatin, a hormone released by the hypothalamus;
- Insulin like growth factor (IGF-1): This hormone is increased by GH, is responsible for most of the effects of GH, and also reduces the amount of GH released (negative feedback);
- High blood sugar (hyperglycaemia);
- Leptin, a hormone produced by fat cells. It signals to the hypothalamus that the body has had enough to eat;
- Excess glucocorticoids, a type of steroid that may be administered as a drug. High levels of glucocorticoid increase the number of GH receptors on cells. These detect GH, so if there are more of them, the action of GH on cells is greater;
- Beta-2-adrenergic receptor activation. These are a type of receptor that are stimulated by hormones released during times of stress, and also by certain drugs such as adrenaline. If these are activated, GH action is decreased.
GH secretion is at its peak (150 µg/kg) during puberty, and declines by about 50% every seven years to about 25 µg/kg by age 55.
What are the actions of growth hormone?
Metabolic effects of growth hormone include:
- Increased breakdown of stored fat (triglyceride), and increased use of this fat to create energy;
- Counteracting the actions of insulin. Insulin moves sugar from the blood into cells, so GH opposes this;
- Retention (holding in the body) of sodium, phosphate and water; and
- Stimulation of protein production (a large component of muscles).
Too much GH leads to the condition known as acromegaly or gigantism. Growth hormone deficiency may occur in a range of conditions.
Recombinant human GH (rhGH) is a form of artificially produced GH, and may be administered as a drug to manage a range of conditions, such as:
- GH deficiency in adults due to hypothalamic pituitary disease;
- Chronic kidney disease leading to short stature (height);
- Short stature due to Turner’s syndrome or Prader-Willi syndrome;
- HIV-associated wasting in adults; and
- Small for gestational age infants who do not reach sufficient height.
Why is growth hormone used to manage obesity?
GH reduces the uptake of free fatty acids by adipocytes (fat storage cells). It inhibits LPL, which controls the buildup of triglycerides in fatty tissue. LPL breaks down triglycerides, releasing free fatty acids (FFAs) which may then be taken up by fat cells. GH also encourages the body to use FFAs as an energy source, and to reduce the use of sugars (glucose) and proteins for energy. The impact of GH in reducing LPL activity is greater in fat deposits within the abdomen than in subcutaneous fat. Other hormones that impact on LDL are insulin (increased activity); and catecholamines (dopamine, etc), testosterone and oestrogen (decreased activity).
The presence of circulating FFA reduces the release of GH by the pituitary. The elevation of FFA in obese individuals (due to greater fatty tissue mass) may be partially responsible for the lower levels of GH evident in obesity. There is also increased clearance of GH in obese individuals, through mechanisms that are not well understood. Also, the increased levels of IGF-1 seen in obese individuals has a negative feedback effect on GH release. Finally, the high levels of insulin seen in many obese individuals reduces the release of GH by the pituitary in response to GHRH.
GH assists the action of HSL, which breaks down stored triglycerides into FFAs and glycerol. This allows the use of FFAs as fuel sources elsewhere in the body. The glycerol portion of the triglyceride must be removed in order to enable FFAs to exit the fat cell. GH increases the number of receptors on the cell that recognise hormones that stimulate HSL activity. It also prevents the breakdown of messengers in the cell that allow the effects of HSL to occur.
GH also has a direct impact on the number and maturity of fat cells. Through IGF-1, GH stimulates production of pre-fat cells; however, GH then prevents them from maturing.
GH opposes skeletal muscle breakdown during fasting. When an individual is eating very few calories, GH encourages the use of fat for energy rather than sugar or proteins. This prevents the breakdown of muscle in order to produce proteins for energy.
How effective is growth hormone in the management of obesity?
On the other hand, many other studies have reported that GH is effective in reducing fat mass, especially visceral fat. These studies were performed on adults with GH deficiency due to pituitary disease, or low levels of GH associated with obesity in the absence of pituitary disease. One review found that “GH replacement with or without diet and exercise interventions effectively reduces visceral adipose tissue (VAT) and improves lipid abnormalities in GH deficient adults.”24 This review concluded that “GH therapy decreases VAT in subjects with adult GH deficiency and in GH-replete viscerally obese adults. It is generally agreed that adults with GHD and visceral obesity should be given GH replacement therapy. The use of GH, with or without insulin sensitisers, represents a novel, albeit expensive, adjunct to diet and exercise in GH-replete obese individuals who are striving to reduce their cardiovascular risk.”24
A further study showed a small but significant reduction in visceral obesity, and an increase in lean mass and body weight with the administration of GH at physiological (normal for the body) doses. GH administration at high levels was not found to be an effective treatment in viscerally obese subjects. Another study demonstrated a 1.6-fold increase in fat loss due to administration of GH compared with placebo, and concurrent increase in lean body mass.
It has been suggested that the negative results seen in some trials may be due to administration of high doses of GH leading to high levels of insulin. This leads to the formation of fat, which may offset the lipolytic effect of the growth hormone.
What are the risks of growth hormone use?
- Insulin resistance (pre-diabetes)
- Swelling
- Joint pain
- Carpal tunnel syndrome
These are reported to be reversible when treatment with GH is stopped.
There have been some concern that GH use may lead to cancer, particularly if it is given in high doses over long periods of time. A 12–35% increase in pre-cancerous polyps in the colon occurs in patients with acromegaly, and colon cancer occurs in 6.9% of cases. However, it has been suggested that individuals with acromegaly are exposed to higher levels of GH over longer periods of time than would occur with therapeutic GH administration.
Another study reported an increased incidence of leukaemia in children treated with human pituitary GH replacement therapy. Subsequent studies did not confirm this increase. A study published in the Lancet found greater risk of death from Hodgkin’s disease, colorectal cancer and cancer overall in patients treated with human pituitary GH. However, in conjunction with other studies, the authors concluded that, if high-risk groups such as those with chromosomal fragility were excluded, leukaemia risk was not substantially raised.
A recent study designed to further examine this issue analysed the impact of daily GH administration to rats over two years and found no growth of cancer. One expert wrote that it is important to monitor the levels of IGF-1 and IGF-1-binding protein-3 (increased by GH) to ensure therapy resulted in age appropriate limits. IGF-1 prevents cell death, and may therefore prevent the death of cells with mutations which may become cancer cells.
More information
 |
For more information on obesity, health and social issues, and methods of weight loss, as well as some useful tools, see Weight Loss. |
References
- Leung KC, Ho KK. Measurement of growth hormone, insulin-like growth factor I and their binding proteins: The clinical aspects. Clin Chim Acta. 2001; 313(1-2): 119-23. [Abstract]
- Hartman ML, Veldhuis JD, Thorner MO. Normal control of growth hormone secretion. Horm Res. 1993; 40(1-3): 37-47. [Abstract]
- Pong SS, Chaung LP, Dean DC, Nargund RP, Patchett AA, Smith RG. Identification of a new G-protein-linked receptor for growth hormone secretagogues. Mol Endocrinol. 1996; 10(1): 57-61. [Abstract]
- Kojima M, Hosoda H, Date Y, Nakazato M, Matsuo H, Kangawa K. Ghrelin is a growth-hormone releasing acylated peptide from stomach. Nature. 1999; 402: 656-60. [Abstract]
- Giustina A, Veldhuis JD. Pathophysiology of the neuroregulation of growth hormone secretion in experimental animals and the human. Endocr Rev. 1998; 19(6): 717-97. [Abstract | Full text]
- Van Cauter E, Leproult R, Plat L. Age-related changes in slow wave sleep and REM sleep and relationship with growth hormone and cortisol levels in healthy men. JAMA. 2000; 284(7): 861-8. [Abstract | Full text]
- Salvatori R. Growth hormone and IGF-1. Rev Endocr Metab Disord. 2004; 5(1): 15-23. [Abstract]
- Unger RH. The hyperleptinemia of obesity-regulator of caloric surpluses. Cell. 2004; 117(2): 145-6. [Abstract | Full text]
- Frank SJ. Growth hormone signalling and its regulation: Preventing too much of a good thing. Growth Horm IGF Res. 2001; 11(4): 201-12. [Abstract]
- Gharib H,Cook DM, Saenger PH, Bengtsson BA, Feld S, Nippoldt TB, et al. American Association of Clinical Endocrinologists medical guidelines for clinical practice for growth hormone use in adults and children – 2003 update. Endocr Pract. 2003; 9(1): 64-76. [Abstract | Full text]
- Richelsen B. Effect of growth hormone on adipose tissue and skeletal muscle lipoprotein lipase activity in humans. J Endocrinol Invest. 1999; 22(5 Suppl): 10-5. [Abstract]
- Oscarsson J, Ottosson M, Edén S. Effects of growth hormone on lipoprotein lipase and hepatic lipase. J Endocrinol Invest. 1999; 22(5 Suppl): 2-9. [Abstract]
- Nam SY, Marcus C. Growth hormone and adipocyte function in obesity. Horm Res. 2000; 53(Suppl 1): 87-97. [Abstract]
- Pombo M, Pombo CM, Astorga R, Cordido F, Popovic V, Garcia-Mayor RV, et al. Regulation of growth hormone secretion by signals produced by the adipose tissue. J Endocrinol Invest. 1999; 22(5 Suppl): 22-6. [Abstract]
- Langendonk JG, Meinders AE, Burggraaf J, Frölich M, Roelen CA, Schoemaker RC, et al. Influence of obesity and body fat distribution on growth hormone kinetics in humans. Am J Physiol. 1999; 277(5 Pt 1): E824-9. [Abstract | Full text]
- Lanzi R, Luzi L, Caumo A, Andreotti AC, Manzoni MF, Malighetti ME, et al. Elevated insulin levels contribute to the reduced growth hormone (GH) response to GH-releasing hormone in obese subjects. Metabolism. 1999; 48(9): 1152-6. [Abstract]
- Clasey JL, Weltman A, Patrie J, Weltman JY, Pezzoli S, Bouchard C, et al. Abdominal visceral fat and fasting insulin are important predictors of 24-hour GH release independent of age, gender, and other physiological factors. J Clin Endocrinol Metab. 2001; 86(8): 3845-52. [Abstract | Full text]
- Iranmanesh A, Grisso B, Veldhuis JD. Low basal and persistent pulsatile growth hormone secretion are revealed in normal and hyposomatotropic men studied with a new ultrasensitive chemiluminescence assay. J Clin Endocrinol Metab. 1994; 78(3): 526-35. [Abstract]
- Bramnert M, Segerlantz M, Laurila E, Daugaard JR, Manhem P, Groop L. Growth hormone replacement therapy induces insulin resistance by activating the glucose-fatty acid cycle. J Clin Endocrinol Metab. 2003; 88(4): 1455-63. [Abstract | Full text]
- Flint DJ, Gardner MJ. Influence of growth hormone deficiency on growth and body composition in rats: Site-specific effects upon adipose tissue development. J Endocrinol. 1993; 137(2): 203-11. [Abstract]
- Björntorp P. Hormonal control of regional fat distribution. Hum Reprod. 1997; 12(Suppl 1): 21-5. [Abstract]
- Wabitsch M, Braun S, Hauner H, Heinze E, Ilondo MM, Shymko R, et al. Mitogenic and antiadipogenic properties of human growth hormone in differentiating human adipocyte precursor cells in primary culture. Pediatr Res. 1996; 40(3): 450-6. [Abstract | Full text]
- Nørrelund H, Nair KS, Jørgensen JO, Christiansen JS, Møller N. The protein-retaining effects of growth hormone during fasting involve inhibition of muscle-protein breakdown. Diabetes. 2001; 50(1): 96-104. [Abstract | Full text]
- Shadid S, Jensen. Effects of growth hormone administration in human obesity. Obes Res. 2003; 11(2): 170-5. [Abstract]
- Lucidi P, Parlanti N, Piccioni F, Santeusanio F, De Feo P. Short-term treatment with low doses of recombinant human GH stimulates lipolysis in visceral obese men. J Clin Endocrinol Metab. 2002; 87(7): 3105-9. [Abstract | Full text]
- Nam SY, Kim KR, Cha BS, Song YD, Lim SK, Lee HC, et al. Low-dose growth hormone treatment combined with diet restriction decreases insulin resistance by reducing visceral fat and increasing muscle mass in obese type 2 diabetic patients. Int J Obes Relat Metab Disord. 2001; 25(8): 1101-7. [Abstract | Full text]
- Bengtsson BA, Edén S, Lönn L, Kvist H, Stokland A, Lindstedt G, et al. Treatment of adults with growth hormone (GH) deficiency with recombinant human GH. J Clin Endocrinol Metab. 1993; 76(2): 309-17. [Abstract]
- Verhelst J, Abs R, Vandeweghe M, Mockel J, Legros JJ, Copinschi G, et al. Two years of replacement therapy in adults with growth hormone deficiency. Clin Endocrinol (Oxf). 1997; 47(4): 485-94. [Abstract]
- Chrisoulidou A, Beshyah SA, Rutherford O, Spinks TJ, Mayet J, Kyd P, et al. Effects of 7 years of growth hormone replacement therapy in hypopituitary adults. J Clin Endocrinol Metab. 2000; 85(10): 3762-9. [Abstract | Full text]
- Gibney J, Wallace JD, Spinks T, Schnorr L, Ranicar A, Cuneo RC, et al. The effects of 10 years of recombinant human growth hormone (GH) in adult GH-deficient patients. J Clin Endocrinol Metab. 1999; 84(8): 2596-602. [Abstract | Full text]
- Johannsson G, Johansson J. Effects of growth hormone replacement in adults. In: Bengtsson BA, Monson JP [eds]. GH Replacement in Adults: The First 5 Years of KIMS. Oxford: PharmaGenesis; 2000, pp. 115-32.
- Salomon F, Cuneo RC, Hesp R, Sönksen PH. The effects of treatment with recombinant human growth hormone on body composition and metabolism in adults with growth hormone deficiency. N Engl J Med. 1989; 321(26): 1797-803. [Abstract]
- Hoffman AR, Biller BM, Cook D, Baptista J, Silverman BL, Dao L, et al. Efficacy of a long-acting growth hormone (GH) preparation in patients with adult GH deficiency. J Clin Endocrinol Metab. 2005; 90(12): 6431-40. [Abstract | Full text]
- Snel YE, Brummer RM, Doerga ME, Zelissen PM, Bakker CJ, Hendriks MJ, et al. Adipose tissue assessed by magnetic resonance imaging in growth hormone-deficient adults: The effect of growth hormone replacement and a comparison with control subjects. Am J Clin Nutr. 1995; 61(6): 1290-4. [Abstract | Full text]
- Johannsson G, Mårin P, Lönn L, Ottosson M, Stenlöf K, Björntorp P, et al. Growth hormone treatment of abdominally obese men reduces abdominal fat mass, improves glucose and lipoprotein metabolism, and reduces diastolic blood pressure. J Clin Endocrinol Metab. 1997; 82(3): 727-34. [Abstract | Full text]
- Lucidi P, Parlanti N, Piccioni F, Santeusanio F, De Feo P. Short-term treatment with low doses of recombinant human GH stimulates lipolysis in visceral obese men. J Clin Endocrinol Metab. 2002; 87(7): 3105-9. [Abstract | Full text]
- Tomlinson JW, Crabtree N, Clark PM, Holder G, Toogood AA, Shackleton CH, et al. Low-dose growth hormone inhibits 11 beta-hydroxysteroid dehydrogenase type 1 but has no effect upon fat mass in patients with simple obesity. J Clin Endocrinol Metab. 2003; 88(5): 2113-8. [Abstract | Full text]
- Taaffe DR, Thompson JL, Butterfield GE, Hoffman AR, Marcus R. Recombinant human growth hormone, but not insulin-like growth factor-I, enhances central fat loss in postmenopausal women undergoing a diet and exercise program. Horm Metab Res. 2001; 33(3): 156-62. [Abstract]
- Attallah H, Friedlander AL, Hoffman AR. Visceral obesity, impaired glucose tolerance, metabolic syndrome, and growth hormone therapy. Growth Hormone Igf Res. 2006; 16(Suppl A): S62-7. [Abstract]
- Toogood AA, Holmes SJ, Shalet SM. Monitoring growth hormone replacement therapy. In: Juul A, Jørgensen JOL [eds]. Growth Hormone in Adults: Physiological and Clinical Aspects. Cambridge: Cambridge University Press; 2000, pp. 186-98. [Book]
- Watanabe S, Mizuno S, Oshima LH, Tsunematsu Y, Fujimoto J, Komiyama A. Leukemia and other malignancies among GH users. J Pediatr Endocrinol. 1993; 6(1): 99-108. [Abstract]
- Swerdlow AJ, Higgins CD, Adlard P, Preece MA. Risk of cancer in patients treated with human pituitary growth hormone in the UK, 1959-85: A cohort study. Lancet. 2002; 360: 273-7. [Abstract]
- Ogilvy-Stuart AL, Gleeson H. Cancer risk following growth hormone use in childhood: Implications for current practice. Drug Saf. 2004; 27(6): 369-82. [Abstract]
- Farris GM, Miller GK, Wollenberg GK, Molon-Noblot S, Chan C, Prahalada S. Recombinant rat and mouse growth hormones: Risk assessment of carcinogenic potential in 2-year bioassays in rats and mice. Toxicol Sci. 2007; 97(2): 548-61. [Abstract | Full text]
- Sperling MA, Saenger PH, Hintz R, Wilson T, Rose SR. Special editorial: Growth hormone treatment and neoplasia – coincidence or consequence? J Clin Endocrinol Metab. 2002; 87(12): 5351-2. [Abstract | Full text]
- Münzer T, Harman SM, Hees P, Shapiro E, Christmas C, Bellantoni MF, et al. Effects of GH and/or sex steroid administration on abdominal subcutaneous and visceral fat in healthy aged women and men. J Clin Endocrinol Metab. 2001; 86(8): 3604-10. [Abstract | Full text]
- de Boer H, Blok GJ, Voerman B, Derriks P van der Veen E. Changes in subcutaneous and visceral fat mass during growth hormone replacement therapy in adult men. Int J Obes Relat Metab Disord. 1996; 20(6): 580-7. [Abstract]
All content and media on the HealthEngine Blog is created and published online for informational purposes only. It is not intended to be a substitute for professional medical advice and should not be relied on as health or personal advice. Always seek the guidance of your doctor or other qualified health professional with any questions you may have regarding your health or a medical condition. Never disregard the advice of a medical professional, or delay in seeking it because of something you have read on this Website. If you think you may have a medical emergency, call your doctor, go to the nearest hospital emergency department, or call the emergency services immediately.







